Twee mijlpalen behaald in de richting van toegang tot kleine boringen met Impella
DANVERS, Massachusetts – (BUSINESS WIRE) – Abiomed (NASDAQ: ABMD) kondigt de verwezenlijking aan van twee mijlpalen in de ontwikkeling van toegang voor kleine boringen voor de Impella-hartpomp. De Impella ECP-hartpomp heeft de eerste fase van haar early feasibility study (EFS) van de Amerikaanse Food en Drug Administration (FDA) voltooid en de FDA heeft 510 (k) goedkeuring verleend aan de Impella XR-huls.
Dit persbericht bevat multimedia. Bekijk de volledige release hier: https://www.businesswire.com/news/home/20201215005395/en/
De Impella XR-huls is een onopvallende huls die uitzet en terugdraait, waardoor kleine boring toegang en sluiting mogelijk is met de Impella 2.5 hartpomp. Het wordt ingevoegd op 10 French (Fr) en de flexibele nitinolvlechten zetten even uit tijdens Impella-levering en trekken zich dan terug, waardoor de toegang voor complexe interventies wordt vereenvoudigd. De Impella XR-sheath is bedoeld om minder trauma te veroorzaken op de arteriële toegangsplaats in vergelijking met sheaths met een grote boring.
Two Milestones Achieved Toward Small Bore Access with Impella
DANVERS, Mass.–(BUSINESS WIRE)– Abiomed (NASDAQ: ABMD) announces the achievement of two milestones in the development of small bore access for the Impella heart pump. The Impella ECP heart pump has completed the first stage in its U.S. Food and Drug Administration (FDA) early feasibility study (EFS) and the FDA has granted 510(k) clearance to the Impella XR sheath.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20201215005395/en/
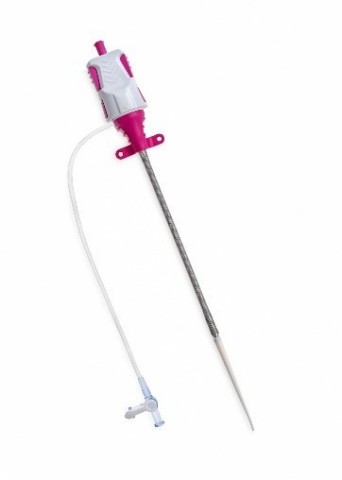
The Impella XR sheath inserts at 10 Fr, momentarily expands during Impella delivery, then recoils. (Photo: Business Wire)
The Impella XR sheath is a low-profile sheath that expands and recoils, allowing for small bore access and closure with the Impella 2.5 heart pump. It inserts at 10 French (Fr) and the flexible, nitinol braids momentarily expand during Impella delivery then recoil, simplifying access for complex interventions. The Impella XR sheath is intended to produce less trauma at the arterial access site compared to large bore sheaths.
The Impella XR sheath has been studied in patients outside of the United States on multiple occasions. The first patient in the U.S. is expected during Q4 of fiscal year 2021.
Additionally, Abiomed has successfully completed the first stage of the EFS of Impella ECP by enrolling and treating five patients. Impella ECP is the world’s smallest heart pump. It measures 9 Fr in diameter upon insertion and removal from the body. Once in the body, it expands to support the heart’s pumping function, providing peak flows greater than 3.5 L/min.
The prospective, multi-center, non-randomized EFS is designed to allow Abiomed, study investigators, and the FDA to make qualitative assessments about the safety and feasibility of Impella ECP use in high-risk percutaneous coronary intervention (PCI) patients. Abiomed will now submit data to the FDA and request to move to a second stage of the study with expanded enrollment.
“These milestones are important steps toward the fulfillment of Abiomed’s vision of creating an innovative portfolio of products that are smaller, smarter and more connected,” said Chuck Simonton, MD, Abiomed’s chief medical officer. “Design innovations like Impella ECP and Impella XR sheath will allow for a smaller access site with Impella, which will continue to drive better patient care and outcomes.”
Impella ECP is available for investigational use only and is not approved for use outside of clinical studies.
ABOUT ABIOMED
Based in Danvers, Massachusetts, USA, Abiomed, Inc. is a leading provider of medical devices that provide circulatory support. Our products are designed to enable the heart to rest by improving blood flow and/or performing the pumping of the heart. For additional information, please visit: www.abiomed.com.
ABOUT IMPELLA XR SHEATH
The Impella XR Sheath set is intended for use for the percutaneous introduction of the Impella 2.5 Catheter and ancillary devices.
ABOUT IMPELLA ECP
CAUTION: Impella ECP is an investigational device, limited by federal law to investigational use only.
ABOUT IMPELLA HEART PUMPS
The Impella 2.5® and Impella CP® devices are U.S. FDA approved to treat certain advanced heart failure patients undergoing elective and urgent percutaneous coronary interventions (PCI), such as stenting or balloon angioplasty, to reopen blocked coronary arteries.
The Impella 2.5, Impella CP, Impella CP with SmartAssist®, Impella 5.0®, Impella LD®, and Impella 5.5® with SmartAssist® are U.S. FDA approved to treat heart attack or cardiomyopathy patients in cardiogenic shock, and have the unique ability to enable native heart recovery, allowing patients to return home with their own heart.
FORWARD-LOOKING STATEMENTS
This press release contains forward-looking statements. Forward-looking statements are subject to risks and uncertainties such as those described in Abiomed’s periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
View source version on businesswire.com: https://www.businesswire.com/news/home/20201215005395/en/
Contacts
Sarah Karr
Communications Manager
skarr@abiomed.com
(978) 882-8211
Todd Trapp
Vice President and Chief Financial Officer
ttrapp@abiomed.com
(978) 646-1680

