MagBio ontvangt 510(k) goedkeuring en CE-markering voor MagXtract-verzamelbuis voor COVID-19-tests
Nu gelanceerd in de VS en Europa als het eerste door de FDA goedgekeurde guanidine-vrije moleculaire transportmedium; Biedt verbeterde veiligheid, stabiliteit en kosteneffectiviteit
GAITHERSBURG, Md.–(BUSINESS WIRE)– MagBio Genomics, Inc., een wereldwijd bedrijf dat zich richt op producten op basis van magnetische kralen voor moleculaire diagnostiek en genomisch onderzoek van vloeibare biopsie, heeft de goedkeuring van de Amerikaanse Food en Drug Administration (FDA) 510(k) aangekondigd, CE-markering en wereldwijde lancering van de MagXtract Collection Tube. De MagXtract Collection Tube wordt gebruikt voor het verzamelen en verwerken van monsters en is het eerste door de FDA goedgekeurde guanidine-vrije moleculaire transportmedium dat beschikbaar is in de VS voor COVID-19-tests. In Europa is de MagXtract Collection Tube veelzijdig inzetbaar voor het testen van zowel COVID-19 als Influenza. Het is ook gevalideerd voor het verzamelen en stabiliseren van bacteriële en schimmelmonsters in onderzoek studies.
Dit persbericht bevat multimedia. Bekijk de volledige release hier: https://www.businesswire.com/news/home/20220912005273/en/
MagBio Receives 510(k) Clearance and CE Mark for MagXtract Collection Tube For COVID-19 Testing
Now Launching in U.S. and Europe as the first FDA-cleared Guanidine-Free Molecular Transport Medium; Offers Improved Safety, Stability and Cost-Effectiveness
GAITHERSBURG, Md.–(BUSINESS WIRE)– MagBio Genomics, Inc., a global company focused on magnetic bead-based products for molecular diagnostics and liquid biopsy genomic research, announced U.S. Food and Drug Administration (FDA) 510(k) clearance, CE Mark approval and global launch of its MagXtract Collection Tube. Used for sample collection and processing, the MagXtract Collection Tube is the first guanidine-free molecular transport medium, FDA-cleared device available in the U.S. for COVID-19 testing. In Europe the MagXtract Collection Tube is versatile, available for testing both COVID-19 and Influenza. It is also validated for bacterial and fungal sample collection and stabilization in research studies.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20220912005273/en/
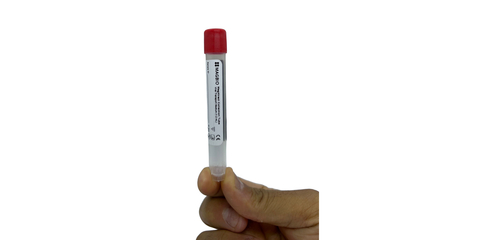
The new MagXtract Collection Tube from MagBio Genomics offers improved safety, stability and cost-effectiveness for sample collection and processing. Now available in the U.S. for COVID-19 testing and Europe for both COVID-19 and Influenza. MagXtract is the first FDA-cleared guanidine-free molecular transport medium that offers direct lysis and room temperature stability of RNA. The FDA has advised laboratory staff at COVID-19 sample processing facilities to avoid collection devices that use guanidine-based mediums because when cleaned with bleach, it creates cyanide gas that is highly toxic and can also be fatal. (Photo: Business Wire)
“Accurate detection of pathogens by molecular assays depends on proper collection and stabilization of samples. Poor sample stabilization results in nucleic acid degradation and overgrowth of some micro-organisms, which may lead to patient misdiagnosis,” said Hyacinth K. Ntchobo, Ph.D., CEO, MagBio Genomics. “The MagXtract Collection Tube addresses these issues by providing cost-effective room temperature sample transportation and nucleic acid stabilization over prolonged time, providing clinical laboratories with high-quality samples for accurate molecular detection of pathogens.”
With the launch of the MagXtract Collection Tube, MagBio offers improved safety, stability and cost-effective sample collection and processing to the worldwide market:
- Safety during disinfection: A guanidine-free, proprietary medium makes it safe for use in clinical settings by eliminating the risk of creating cyanide gas during cleaning, which is highly toxic and can be fatal. The FDA issued a letter to laboratory staff at COVID-19 sample processing facilities to avoid collection devices that use guanidine.
- Sample Handling and Processing Safety: The medium directly lyses cells during transport and inactivates pathogens, eliminating the need for containment use.
- Stability: Ambient temperature stability of RNA samples for up to eight days eliminates the need for cold storage/shipping.
- Efficiency and cost-effectiveness: There is no need to use a lysis buffer because the transport medium also acts as a lysis buffer, reducing costs in nucleic acid extraction workflow.
To learn more about the MagXtract Collection Tube or to request a sample pack, visit www.magbiogenomics.com.
About MagBio Genomics, Inc.
MagBio Genomics is a global company that develops and commercializes magnetic bead-based products for nucleic acid isolation, including biomarkers as tools for molecular diagnostics and liquid biopsy genomic research. The current focus encompasses products that cover complete sample preparation including safeguarding integrity of bio-samples, efficient isolation of nucleic acids and circulating biomarkers to target enrichment from biological samples. Our products are applied to human genetic research, including molecular diagnostics, cancer genomic studies and noninvasive prenatal testing. “We simply make NGS better.” For more information, visit www.magbiogenomics.com and follow the company on Twitter and LinkedIn.
View source version on businesswire.com: https://www.businesswire.com/news/home/20220912005273/en/
Contacts
Joni Ramirez
Merryman Communications
joni@merrymancommunications.com
323-532-0746

