Teckro lanceert mobiel platform voor klinische proeven van de volgende generatie, waardoor sitepersoneel sneller antwoorden krijgt met minder klikken
De lancering komt op het moment dat het bedrijf zijn nieuwe “Teckro Powered”-programma voor sites onthult om de acceptatie van moderne proefpraktijken te stimuleren
LIMERICK, Ierland–(BUSINESS WIRE)– Teckro, de maker van het enige platform voor klinische proeven dat samenwerking en realtime besluitvorming tussen en tussen belanghebbenden mogelijk maakt, kondigt nieuwe productmogelijkheden aan om de acceptatie van moderne proefpraktijken op de site te versnellen, waardoor de communicatie zelfs makkelijker in de zorg. Een mobiele app van de volgende generatie geeft onderzoekers en locatiemedewerkers antwoorden in minder klikken, terwijl platformverbeteringen ertoe leiden dat nieuwe onderzoeken in de helft van de tijd operationeel zijn op Teckro.
Met het oog op zijn visie om elke arts bij klinisch onderzoek te betrekken, onthult Teckro ook zijn “Teckro Powered” site-programma om een gemeenschap van onderzoekssites op te zetten die klaar zijn om de industrie vooruit te helpen en toekomstige productvereisten vorm te geven. Tegenwoordig vertrouwen meer dan 23.000 onderzoekslocaties over de hele wereld op het Teckro-platform voor klinische proeven op alle therapeutische gebieden.
“Met een kritische massa van locaties die Teckro gebruiken, verdiepen we onze toewijding aan een eenvoudige, locatiegestuurde ervaring waarmee artsen gemakkelijk aan onderzoek kunnen deelnemen zonder de tijd voor patiëntenzorg in gevaar te brengen”, zegt Teckro CEO Gary Hughes. “Sponsors kunnen de complexiteit van klinische onderzoeken verlichten door op een vlotte manier te communiceren met locaties, ook wanneer onderzoekers directe begeleiding kunnen krijgen van onderzoeksexperts op het punt van zorg.”
Teckro Launches Next-Generation Clinical Trial Mobile Platform, Giving Site Staff Faster Answers in Fewer Clicks
The launch comes as the company unveils its novel “Teckro Powered” program for sites to boost adoption of modern trial practices
LIMERICK, Ireland–(BUSINESS WIRE)– Teckro, the creator of the only clinical trials platform that facilitates collaboration and real-time decision-making between and among stakeholders, announces new product capabilities to accelerate site adoption of modern trial practices, making communication even easier at the point of care. A next-generation mobile app gives investigators and site staff answers in fewer clicks, while platform enhancements will result in new studies operational on Teckro in half the time.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210929005472/en/
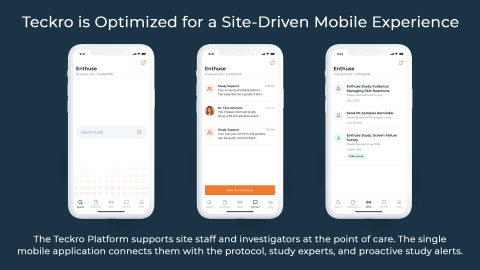
(Graphic: Teckro)
Toward its vision of engaging every physician in clinical research, Teckro also unveils its “Teckro Powered” site program to establish a community of research sites ready to advance the industry and help shape future product requirements. Today, more than 23,000 research sites around the world rely on the Teckro platform for clinical trials in all therapeutic areas.
“With a critical mass of sites using Teckro, we deepen our commitment to a simple, site-driven experience that allows physicians to easily engage in research without compromising time caring for patients,” says Teckro CEO Gary Hughes. “Sponsors can ease the complexity of clinical trials with a frictionless way to communicate with sites, including when investigators can receive direct guidance from study experts at the point of care.”
First-Of-Its-Kind Site Program: Teckro Powered
“Teckro Powered” is a novel program open to any research site. The program includes a dedicated sandbox to preview Teckro capabilities and input into future requirements. In addition with the program, sites and site networks can:
- Build a technology-ready brand. Teckro Powered sites are technically savvy. Designated with a program badge on their websites and social media platforms, sites can use this to differentiate themselves and build their reputations as “sites of choice” for sponsors.
- Expand physician participation in research. Behind the Teckro Powered program is a community of research sites to open a new way to collaborate and advance the industry. With the strength of a Teckro Powered community, the goal is to encourage more physicians to participate in clinical trials, reaching broader, diverse patient populations.
- Limit impact of staff turnover. Teckro Powered is a way for sites to mitigate disruption from staff turnover. With study resources conveniently hosted in Teckro and accessible from a smartphone, sites can simplify new study member onboarding. A central source for content, communication and collaboration reduces the time to train new staff over the course of a trial.
“The Teckro Powered program fills a gap in the industry,” says Director of Site Strategy Silvina Baudino. “Working with sites for almost 20 years, I know how challenging clinical research can be. Teckro is dedicated to the site experience, which you see in both the new product capabilities and the investment in the Teckro Powered program. We are creating a way for sites to differentiate themselves and participate in a community of research sites.”
Maximizing Sponsor-Site Collaboration
Teckro’s latest platform enhancements go toward solving gaps in communication and easing the burden on site staff. A hub for study content and communication, the new Teckro capabilities include:
- Next-generation mobile application. The streamlined app provides a simple, mobile-first user experience to support point of care decisions. Based on deep user research, the new mobile application is optimally designed for clinical trial answers in just a couple of clicks. The new Teckro mobile app is now available in the app stores for Apple and Android smartphones.
- Diverse experts on demand. Teckro is expanding its communication to support more granular groups of experts for different types of site queries. Real-time guidance from those best suited to provide answers eliminates the need for a “switchboard” or for study monitors to operate as a go-between. The feature will be available in the fourth quarter.
- Increased speed and scale. Underlying platform improvements mean new studies can be added in half the time. This allows sponsors to standardize on Teckro as a resource for their sites to access current study content and communications across trials.
Meet Teckro at the Global Site Solution Summit
Teckro is a sponsor at the upcoming Global Site Solution Summit (October 1-3). Visit us at Booth 716 to see a demo and talk with our experts. You can also request a meeting with Silvina Baudino (Teckro director of site strategy) or Malia Lewin (Teckro global head of strategy) by completing the form on our event page. Silvina will also participate as a panelist in the breakout session “It’s Not You, It’s Me: How to Manage Long-Term Partnerships” on both October 2 at 1:30 p.m. and October 3 at 9 a.m.
About Teckro
Recognized as one of the fastest growing clinical trial start-ups, Teckro transforms the study protocol into a hub of communication and collaboration. It’s a digital platform that connects all study stakeholders with the knowledge they need, when they need it. Thousands of investigators and more than 23,000 sites around the world rely on Teckro to execute clinical trials from biopharma sponsors, including 12 out of the Top 20 pharma companies. Teckro supports all therapeutic areas, all phases, and any trial format ranging from traditional in-clinic, decentralized clinical trials, or a hybrid approach.
More Information
- Visit teckro.com
- Connect with Teckro on LinkedIn
- Follow @teckroofficial on Twitter and Instagram
- Like Teckro on Facebook
- Subscribe to the Teckro YouTube channel
- Named as one of Linkedin’s official top ten Irish startups 2021
- Featured on Sifted’s Irish startups to watch list 2021
View source version on businesswire.com: https://www.businesswire.com/news/home/20210929005472/en/
Contacts
Hannah Lippitt
hannah.lippitt@teckro.com
+447393334501

