Vascudyne kondigt succesvol eerste menselijk gebruik aan van ECHT vaattransplantaat voor toegang tot hemodialyse
–News Direct–
Vascudyne, Inc. kondigde vandaag het succesvolle eerste gebruik bij mensen van zijn TRUE™-vasculaire graft aan bij patiënten met nierziekte in het eindstadium die toegang tot hemodialyse nodig hebben. De eerste klinische procedures werden uitgevoerd door Adrian Ebner, MD, directeur van endovasculaire en cardiovasculaire chirurgie bij Sanatorio Italiano in Asuncion, Paraguay, als onderdeel van een vroege haalbaarheidsstudie.
“We zijn verheugd om samen te werken met Dr. Ebner en zijn bekwame team bij Sanatorio Italiano en zijn dankbaar voor hun inzet voor de nieuwste innovaties op het gebied van cardiovasculaire geneeskunde en voor de succesvolle first-in-human TRUE Vascular Graft-implantatieprocedures bij 5 patiënten,” zei Vascudyne CEO Kem Schankereli.
Vascudyne Announces Successful First Human Use of TRUE Vascular Graft for Hemodialysis Access
–News Direct–
Vascudyne, Inc. announced today the successful first-in-human use of its TRUE™ Vascular Graft in end-stage renal disease patients requiring hemodialysis access. The first clinical procedures were performed by Adrian Ebner, MD, Director of Endovascular and Cardiovascular Surgery at Sanatorio Italiano in Asuncion, Paraguay, as part of an early feasibility study.
“We are excited to work with Dr. Ebner and his skilled team at Sanatorio Italiano and are grateful for their commitment to the latest cardiovascular medicine innovations and for the successful first-in-human TRUE Vascular Graft implantation procedures in 5 patients,” said Vascudyne CEO Kem Schankereli.
“Until now, hemodialysis patients who are not candidates for autologous fistulas often receive synthetic grafts, like those made with ePTFE,” said Dr. Ebner. “Synthetic grafts have long-term patency issues with infection, intimal hyperplasia, and thrombosis often leading to failure. All this contributes to patient morbidity and mortality and to the high cost of dialysis care.”
“Vascudyne’s TRUE Vascular Graft with the potential to regenerate and repair promises to have a major impact on the care of dialysis patients,” added Dr. Ebner. “Improved patency and fewer interventions with a completely biological tissue could significantly reduce the overall cost of dialysis care and improve quality of life for patients.”
“The TRUE Vascular Graft handled well and provided excellent flow immediately after implantation,” continued Dr. Ebner. “The device can be felt under the skin which will help with dialysis. Initial follow up showed normal healing and improved flow. There is a tremendous medical need for hemodialysis access solutions, and Vascudyne’s TRUE Vascular Graft has shown very promising initial results.”
“It’s immensely rewarding to see this technology become clinical reality by the Vascudyne team,” said Robert Tranquillo, PhD, Distinguished McKnight University Professor, University of Minnesota. “Decades of research in my lab at the University of Minnesota developing and fine tuning the engineered tissue tube technology have come to fruition and now offer a completely biological vascular graft for patients. I am very excited about Vascudyne’s TRUE™ Tissue technology and all the potential benefits it can bring to patients and look forward to providing ongoing support to the team as they broaden the technology’s clinical applications.”
“Vascudyne is a fast-moving startup with a very successful beginning toward proving clinical relevance,” said Vascudyne General Manager Rick Murphy. “We are a dedicated team of scientists who are committed to innovating solutions with our TRUE Tissue technology for highly demanding medical applications. We are planning additional clinical studies in the next 12 months and will be submitting for market acceptance in the years ahead. We have several more product designs in development and are pursuing ways to accelerate these other programs into the clinical stage. We believe the right material with the correct strategy will help millions of patients across the globe live better lives.”
Vascudyne licensed its proprietary TRUE Tissue technology developed by Professor Robert Tranquillo and his colleagues from the University of Minnesota in 2017.
The TRUE Vascular Graft is not available for commercial sale.
About Vascudyne
Headquartered in the heart of Medical Alley in Minnesota, Vascudyne is on a mission to improve patient care with regenerative biomaterials that are inspired by nature. Vascudyne, a privately held company founded in 2014, uses the TRUE™ Tissue technology to develop TRUE to Nature™ biomaterials for soft tissue repair and replacement. For more information, please visit https://www.vascudyne.com/.
About TRUE Tissue Technology
TRUE™ Tissue is developed from cells isolated from donor tissue and is 100% biological. There are no synthetic materials or chemical fixation used, and implanted tissues are completely cell-derived and acellular. The TRUE Tissue technology can be readily shaped into tubes, sheets, and other geometries making it suitable for many soft tissue applications, is mechanically comparable to native tissues, and is a ready to use, off-the-shelf allograft.
Forward Looking Statements
This announcement contains forward-looking statements. Such statements may include, without limitation, statements identified by words such as “projects,” “may,” “will,” “could,” “would,” “should,” “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “potential” or similar expressions. These statements relate to future events or Vascudyne’s clinical development programs, reflect management’s current beliefs and expectations and involve known and unknown risks, uncertainties and other factors that may cause Vascudyne’s actual results, performance or achievements to be materially different. Vascudyne undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future presentations or otherwise, except as required by applicable law.
IMAGES

Vascudyne TRUE™ Graft First in Human Team
Vascudyne TRUE™ Graft
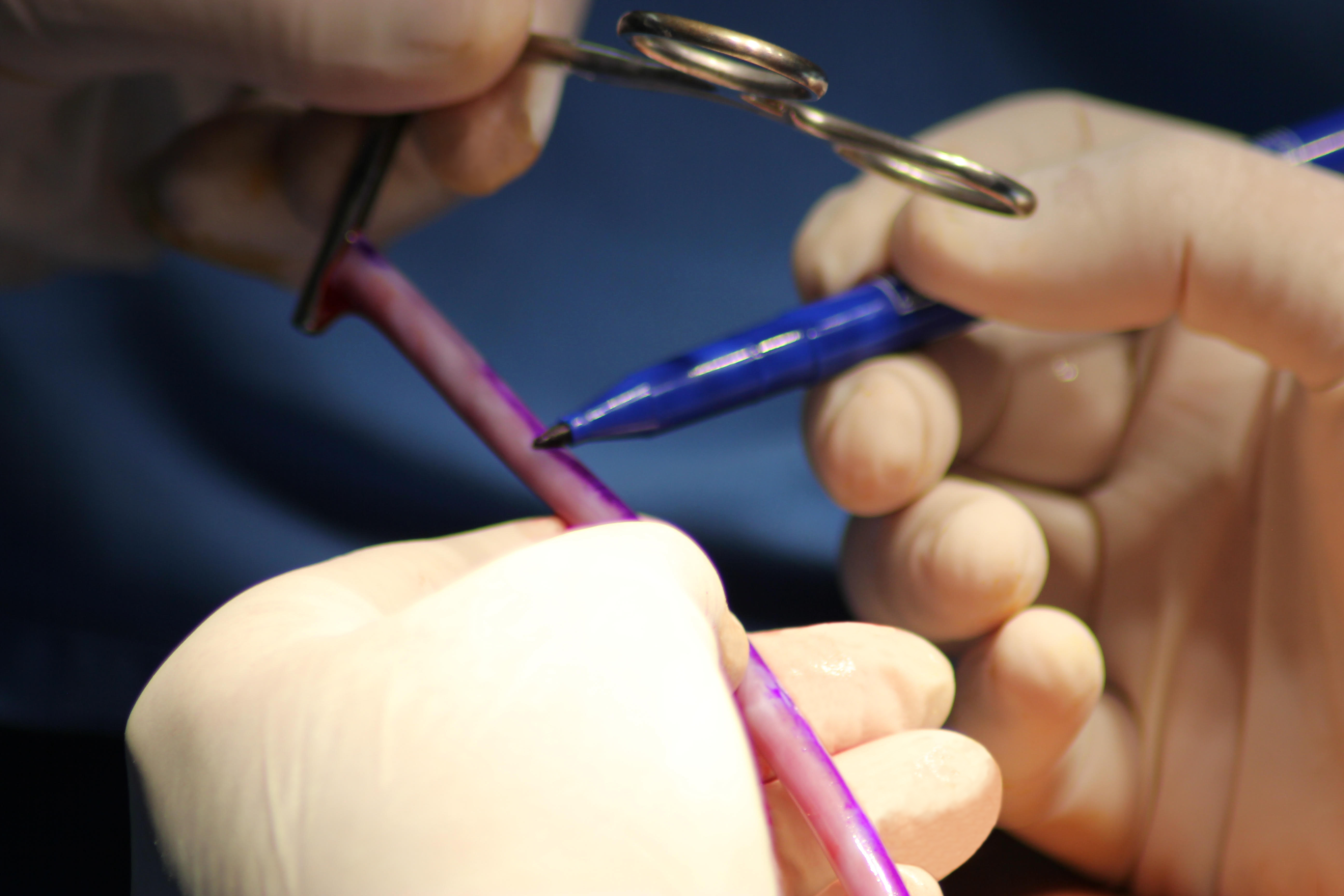
Vascudyne TRUE™ Graft Closeup
Contact Details
Sandy Williams
Marketing Director
Company Website
View source version on newsdirect.com: https://newsdirect.com/news/vascudyne-announces-successful-first-human-use-of-true-vascular-graft-for-hemodialysis-access-352264410
Check out our twitter: @NewsNovumpr
